
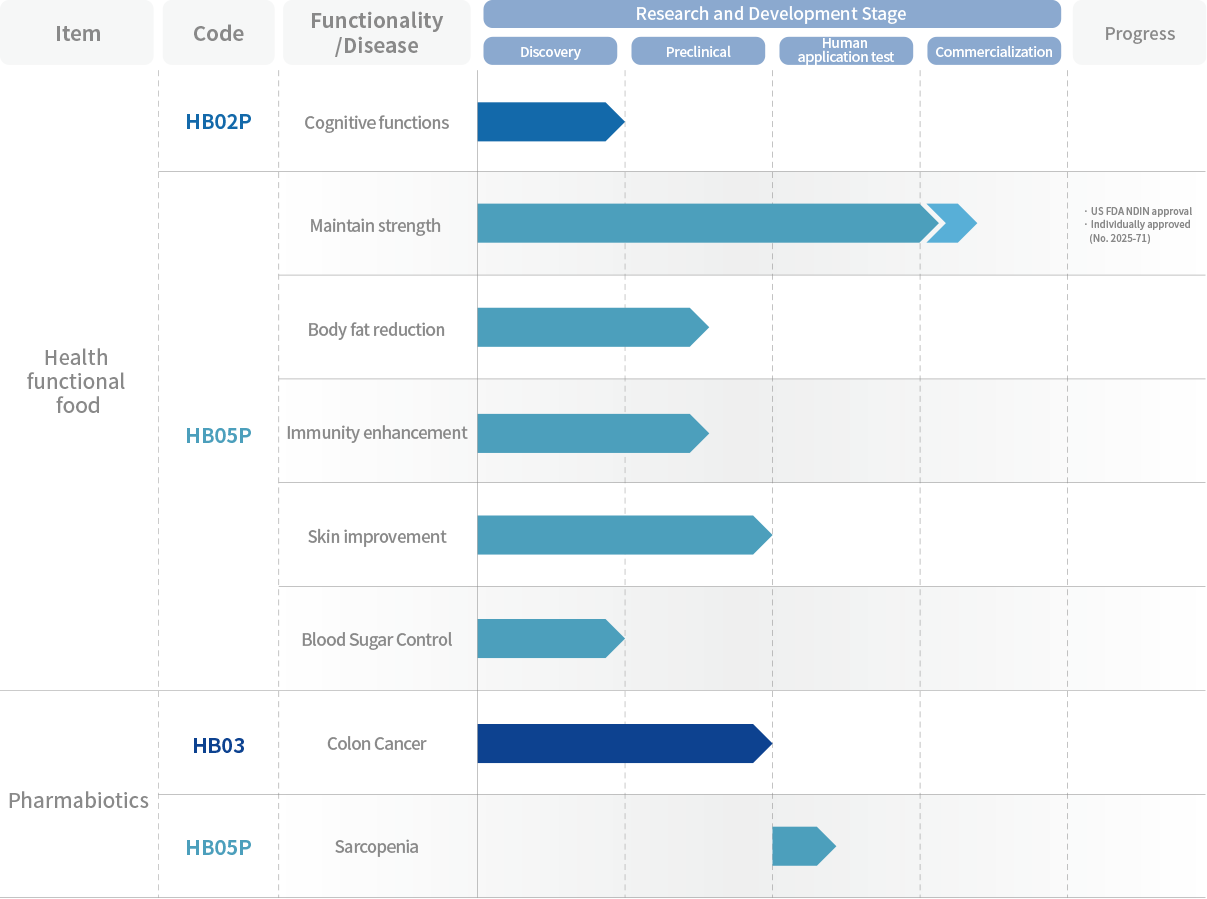
BUSINESS
health through continuous research and development
to the promotion of human health through
continuous research and development
Akkermansia muciniphila Human intestinal strict anaerobic bacterial strain
Metabolic diseases
Degenerative brain
diseases
Cancer

- Gram-negative, non-motile
- Human intestinal commensal bacteria
(1~4% of stool mass, found in more than 75% people) - The first bacterium inoculated in the intestine during infancy
- In the process of global patent application and registration
- Non-(antibiotic resistance, antibacterial material production, pathogenic, toxin-related) genes
- Non-gene transfer related plasmid, non-(mobile, prophage) genes
- Anti-cancer & cognitive function improvement effects
Agathobaculum butyriciproducens Human intestinal strict anaerobic bacterial strain
Degenerative brain
diseases
Autism

- Gram-positive, non-motile
- Human intestinal commensal bacteria
- The world’s first new species separated by HealthBiome Inc.
- Excellent efficacy in treating and preventing degenerative brain diseases (Alzheimer’s disease, Parkin’s disease)
- 1) Report on the effect of treating Alzheimer model (Go et al. 2021)
- 2) Patent application of dementia and depression (US, EU, etc.)
- In the process of global patent application and registration
Akkermansia muciniphila Human intestinal strict anaerobic bacterial strain
Inflammatory Bowel Disease

Revolutionary Postbiotic for Muscle Strength Improvement
Discover HealthBiome's breakthrough pasteurized Akkermansia muciniphila strain isolated from maternal milk of healthy Korean women, clinically proven to improve muscle strength.
One of the most abundant members of the human gut microbiome, heralded as a next-generation probiotic agent due to its various roles in host health.
Not only live strain but also heat-killed strain of A. muciniphila is known to maintain health benefits, making it ideal for stable supplement formulations.
Scientific literature confirms A. muciniphila is naturally present in human milk, suggesting its importance in early human development.

Sarcopenia, the continual loss of muscle mass and functions due to aging, represents one of the most widespread health problems in elderly populations worldwide. This condition significantly impacts quality of life and independence.
Until now, there are no medication approved for prevention or treatment of sarcopenia.
As a dietary supplement, HB05P has proven its efficacy in muscle strength improvement as well as the safety in various non-clinical and clinical studies. We believe the product will serve as an effective solution to mitigate such muscle challenge.
Based on scientific evidence that A. muciniphila is present in human milk, our research team collected breast milk samples from 30 healthy Korean mothers to isolate a novel strain
- Source
- Breast milk samples carefully collected from 30 healthy Korean mothers under strict IRB protocols.
- Isolation
- Advanced microbiological techniques used to isolate the specific A.muciniphila strain.
- Deposit
- The isolated strain was deposited to Korean Culture Center of Microorganism as a patented A. muciniphila strain of HB05 (KCCM12424P).
- Pasteurization
- HB05 undergoes pasteurization after cultivation to create HB05P, a postbiotics, maintaining beneficial properties while ensuring stability.
Our rigorous scientific approach ensures that HB05P is both safe and effective in muscle strength improvement. It began with laboratory studies and progressed through animal safety testing to human clinical trials, all conducted according to strict scientific and regulatory standards.
A placebo-controlled randomized clinical trial involving 80 subjects aged over 60 has demonstrated that oral administration of HB05P (3.0X10^10 cells/day) significantly improves multiple aspects of muscle strength:
Participants showed significant improvements in leg muscle strength (flexor and exensor in left and right leg, P<0.01).
Participants showed significant improvement in leg muscle power (flexor in left and right leg, P<0.05).
Improved hand strength (right hand, P<0.05), a key indicator of overall muscle health.
No SAE (serious adverse event) and HB05P-related AE were reported
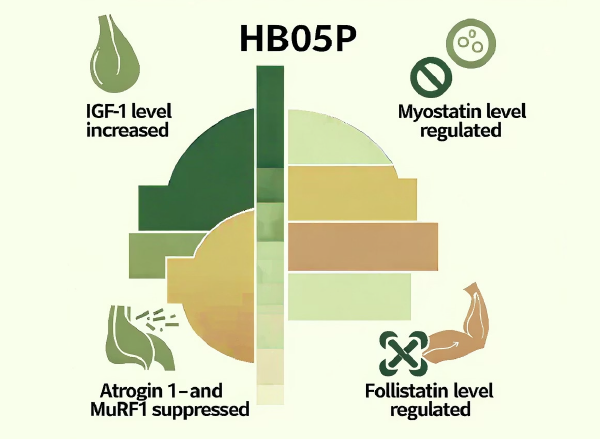
The non-clinical and clinical studies revealed that HB05P works by modulating key hormones involved in muscle anabolism and catabolism:
- IGF-1 (Insulin-like Growth Factor 1) which promotes muscle growth and repair
- Myostatin, a protein that inhibits muscle cell growth and differentiation
- Prealbumin, a sensitive biomarker for short-term protein synthesis and nutritional status
- Follistatin which binds to and inhibits myostatin, promoting muscle growth
- Atrogin-1 and MuRF1, crucial proteins in muscle catabolism (non-clinical)
HB05P has received a no-objection letter from the US FDA for its New Dietary Ingredient Notification (NDIN) application, making it the first Akkermansia muciniphila strain to achieve this safety recognition (NDI 1438).
HB05P completed Individual Recognition process with the Korean Ministry of Food and Drug Safety (MFDS) for approval as a health functional food with functional claim of "May help to maintain muscle strength which can be affected by aging" (Approval No 2025-71).

HB05P represents a significant advancement in addressing age-related muscle decline. As the global population ages, solutions that help maintain muscle function and strength become increasingly vital.
HealthBiome continues to explore additional applications of HB05P as well as to exploit health benefits of human commensal microbiome in its own collection in the strain library.
